/cloudfront-us-east-1.images.arcpublishing.com/gray/TOHKMICXG5FKXBX3YBOGPS52LE.jpg)
Priya Sampathkumar, infectious disease specialist at the Mayo Clinic, tells. This is trying to give us immunity against a different version of the virus,” Dr. “The new vaccine is targeting subtypes of the omicron variant that not all of us have been exposed to yet. Pfizer a lso a nnounced that its reformulated vaccine produces an antibody response against the various omicron sublineages in circulation. Moderna said its updated COVID-19 vaccine prompts a strong immune response against BA.2.86 and generates neutralizing antibodies against other variants, including EG.5, and FL.1.5.1. However, new data suggests that BA.2.86 may not be as immune-evasive as initially thought, and that the updated COVID shots will likely still be effective against the mutated strain. 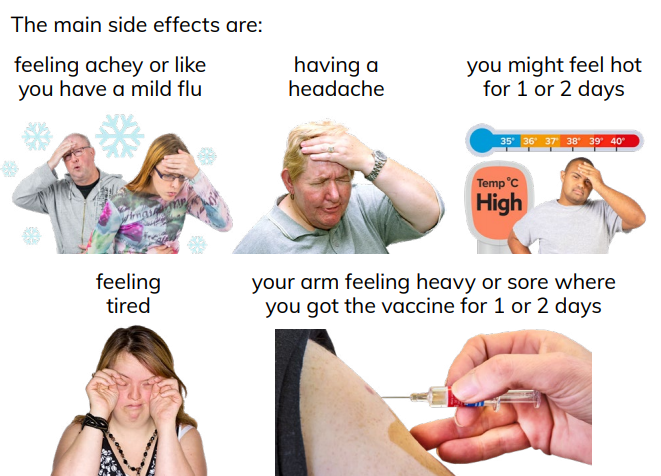 
This summer, a highly mutated new variant called BA.2.86 or “Pirola” gained global attention after health experts predicted it may be better able to bypass immunity from vaccination and prior infection. The updated vaccines are expected to provide good protection against the variants currently circulating, the FDA said. In recent months, the XBB.1.5 subvariant targeted in the updated vaccine has been overtaken by new strains including the EG.5 or "Eris" subvariant, F.L.1.5.1 (also dubbed "Fornax"), and XBB.1.16 or " Arcturus" - which are all descendants of the omicron XBB lineage and close relatives of XBB.1.5, reported previously. The bivalent vaccine is no longer authorized by the FDA, the CDC said. The previous vaccine rolled out last fall w as bivalent, meaning it included two strains - the original SARS-CoV-2 virus and the omicron BA.4 and BA.5 subvariants, says Mulligan. Unlike the last round of boosters, the updated mRNA vaccines are monovalent, which means they target a single variant. Mark Mulligan, director of NYU Langone Vaccine Center, tells. It may be first of what will become an "annual COVID-19 shot" that gets revamped every year, similar to the seasonal influenza vaccine, Dr. for most of 2023.Īlthough many are referring to the updated vaccine as a booster, the shot looks different from its predecessors. The newest COVID-19 booster is an updated vaccine that targets the omicron XBB.1.5 variant, which was the dominant strain circulating in the U.S. Here's what else we know about the updated vaccine, availability, side effects and cost so far. Many parents say that they haven't been able to get their young children vaccinated as pharmacies and pediatrician's offices struggle to secure enough child-size doses.ĭespite these early obstacles, experts say they do not expect long-term, major delays in COVID-19 vaccine availability this fall, and the Biden administration says insurance issues for COVID-19 vaccines have largely been resolved. In recent weeks, some people seeking the updated vaccine have run into high demand, supply delays and insurance headaches, the Associated Press reported. However, the initial phase of the booster rollout has had challenges. “COVID continues to pose a health threat, but we have more and more tools,” Cohen said. Mandy Cohen said at the 2023 National Foundation for Infectious Diseases annual news on Sept. “We fully expect, based on what we’ve seen in previous years, that we’re going to see an increase in COVID (this fall and winter),” CDC Director Dr. The vaccine rollout comes as officials brace for a potential tripledemic wave of COVID-19, influenza and respiratory syncytial virus (RSV) for the second year in a row. 
is just now entering its respiratory virus season, COVID-19 infections and hospitalizations have been on the rise since the summer. The CDC is recommending all Americans who are eligible get least one dose of the updated vaccine to protect against potentially serious outcomes of COVID-19 this fall and winter.Īlthough the U.S. On Tuesday, the FDA approved a third option, a protein-based non-mRNA COVID-19 vaccine from Novavax, for everyone ages 12 and older, NBC News reported. The CDC swiftly endorsed the shots for everyone 6 months and older. Food and Drug Administration authorized two updated mRNA vaccines for 2023 to 2024 from drugmakers Moderna and Pfizer-BioNTech. The reformulated vaccines should also provide good protection against more recent strains, including EG.5 and BA.2.86, according to experts. Unlike the last booster, the new COVID-19 shots are designed to target the omicron XBB.1.5 variant. Centers for Disease Control and Prevention signed off on the rollout of the updated vaccines three weeks ago, millions of doses have arrived at pharmacies, doctor’s offices and clinics around the country. Amid concerns about winter COVID-19 surge in the United States, health officials are urging everyone ages 6 months and older get the new COVID-19 booster this fall.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
